Diagnosing cancer earlier is crucial to saving more lives. Following the recent news of a new genetic test that can predict cancer up to 13 years before the disease develops, Dan Brewer, Visiting Worker in EI’s Vertebrate & Health Genomics group tells us about how the length of telomeres, the caps at the end of the chromosomes which protect the DNA, can be used as a warning sign for the disease.
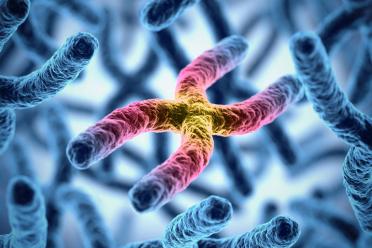
Telomeres are regions of repeated genetic code that occur at the ends of human chromosomes, which help protect the chromosome and provide genetic stability.
The length of telomeres is shortened each time a cell divides and is widely known as a marker for ageing. Shortening can be accelerated by environmental factors such as smoking, physical inactivity and poor diet, which can increase inflammation and oxidative stress.
Short telomeres often induce cell death via apoptosis or senescence, but if the telomeres are critically short the genome becomes unstable, inducing changes such as chromosomal rearrangements and gain and loss of regions.
These changes are necessary to induce cancer development but at the same time a tumour needs to circumvent cell death to thrive. Telomeres have been an active area of research in the cancer field, but little is understood about the balance between these two competing forces that occur with short telomere length.

Interestingly, they found that the rate of age-related telomere length shortening was significantly increased in patients that went on to develop cancer.

Increased risk
In recent work¹ results were reported from the first prospective study of telomere length with multiple measurements: 792 participants from the Normative Aging Study were recruited to provide blood samples at clinical examinations every 3-5 years between 1999 and 2012.
The telomere length of peripheral blood leukocytes were measured by quantitative real-time PCR (polymerase chain reaction) to provide a time course of telomere length for each participant.
213 participants were removed from further analysis as they have been previously diagnosed with cancer. Of the remaining participants, 23 per cent developed cancer during the study. Telomeric length prior to diagnosis was compared between those participants that developed cancer and those that didn’t.
Interestingly, they found that the rate of age-related telomere length shortening was significantly increased in patients that went on to develop cancer. This is suggestive that individuals with a higher rate of telomere shortening, due to genetic or environmental factors, will have an increased risk of cancer development in their life-time, especially early on. Diagnosing cancer earlier in these patients will help to save more lives.

In the majority of cancer types, telomere lengthening mechanisms are hijacked to avoid cell death

Cancer biomarker
Strikingly though, the authors also found that the dynamics of telomere shortening varied as the patient approached diagnosis.
The rate of shortening started to decrease seven years prior to diagnosis reaching a minimum three to four years pre-diagnosis. This suggests that telomere lengthening mechanisms have been activated due to cancer initiation.
In previous research it has been shown that telomere shortening is essential for the initiation of carcinogenesis and that in the majority of cancer types, telomere lengthening mechanisms are hijacked to avoid cell death and hence increase the lifespan of malignant cells.
This mainly occurs through the activation of telomerase, an enzyme that promotes the addition of telomere repeats to the chromosome ends. This is beautifully shown in this study, where an increased shortening rate promotes cancer, but when a tumour has reached a certain stage in its development telomere elongation mechanisms are initiated to stabilise the tumour.
Although this is a small study, there is a strong suggestion that regular measurements of telomere length may be used as a biomarker of both risk of development of and the initiation of cancer, opening up the potential for new ways of diagnosing cancer earlier.

How do you see it working alongside current diagnosis and treatment methods?

Important insight
Despite the small numbers in this study they still showed a positive association between time course measurements of telomere lengths and an increased risk of being diagnosed with prostate cancer at an older age, with the hint that this could extend to other cancers.
Telomere length has been traditionally measured by PCR-based approaches but recently it has become clear that next-generation genome sequencing methods could be used and this may provide a more precise, reliable measure.
This study looking at the time course of telomere length prior to diagnosis of cancer, has provided an important insight into the mechanisms of cancer development and provided further support for the use of telomere length of a marker for both risk and initiation of cancer.
Larger, multicentre trials would be needed to firmly establish telomere length as a useful biomarker.
Diagnosing cancer earlier is obviously something which will benefit millions of people across the world, but what do you think of this research with telomeres? Do you think it will have an impact? How do you see it working alongside current diagnosis and treatment methods? Let us know in the comments section below.
Dr Daniel Brewer is the Senior Bioinformatics Officer at the Cancer Genetics Norwich Medical School in the University of East Anglia.
Blood Telomere Length Attrition and Cancer Development in the Normative Aging Study Cohort
(1) Hou et al, EBioMedicine